INTRODUCING.
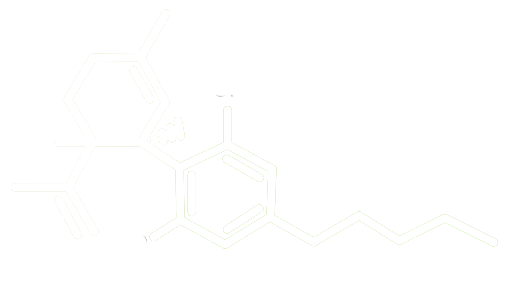
THE SCIENCE OF CANNABINOIDS
Cannabinoids have an extensive history dating back thousands of years, and currently there are thousands of peer-reviewed scientific publications that document the underlying biochemical pathways that cannabinoids modulate.
Learn More

WHAT WE DO
At Cannabis Science, we use an inquiring approach to discover and develop novel cannabinoid-based therapies to improve patients’ lives. From the initiation, our founders have been committed to fostering and maintaining...
Learn More
Latest News.
Cannabis Science Issues 2019 Guidance Report; Under SEC Sanctions the Company Goes Private as It Enters Into Step (2) of Its Growth Transition, Enabling Full Transparency and Revenue Growth
Cannabis Science Enters Transition Stage as Trading of the Company’s Shares is Halted; The Company Will Continue to Implement Its Growth Strategy and Resolve All Outstanding Fully Reporting Requirements
Cannabis Science Partners With CDN Firm NHS Industries to Launch the CBIS CDN Economic Development Plan; CBIS to Use Its Relations With the Harvard International Phytomedicines and Medical Cannabis Institute and Other Leading Institutions
